

P2.6: Towards precision treatments for neuroblastoma
Project Overview
Neuroblastoma (NB) is the most common extracranial solid childhood tumour. While low and intermediate risk patients have a good prognosis with >90% 5-year survival, high risk patients (20-30% of all patients) have much lower survival rates of 40%. High-risk patients are treated with intensive genotoxic combination chemotherapy and radiation therapy, but a large proportion of patients relapse, leaving them with limited or no treatment options. Tragically for survivors, the genotoxic therapies can have severe long-term side effects including hearing loss, secondary cancers, infertility, and endocrine deficiencies. Thus, new types of treatments for these patients are urgently needed. Immunotherapy is an option, but the highly immunosuppressive NB tumour microenvironment limits response rates. Recent studies suggest that relapsed NB features actionable genetic aberrations that could be addressed by targeted therapies. In this project, we are aiming to develop targeted, non-genotoxic therapies for high-risk NB that retain or enhance efficacy, while avoiding the severe long-term side effects of genotoxic treatments.
The project is driven by the combined expertise of the academic and charity partner. Prof. Kolch leads the Systems Biology Ireland group, a world-leading research group in computational modelling and precision medicine. CHI have established translational research resources at the Children’s Clinical Research Unit, and a Biosample Storage Facility, both of which will be accessible to POI. These resources, combined with the use of patient samples collected by our collaborator Dr Cormac Owens via the neuroblastoma sample collection housed in CHI at Crumlin, allow us to apply computational modelling close to the paediatric patient.
We are tackling this problem using two distinct approaches: i) computational modelling for a systematic approach to identifying non-genotoxic NB therapies, and ii) multi-omics approaches to investigate and validate new NB drug targets.
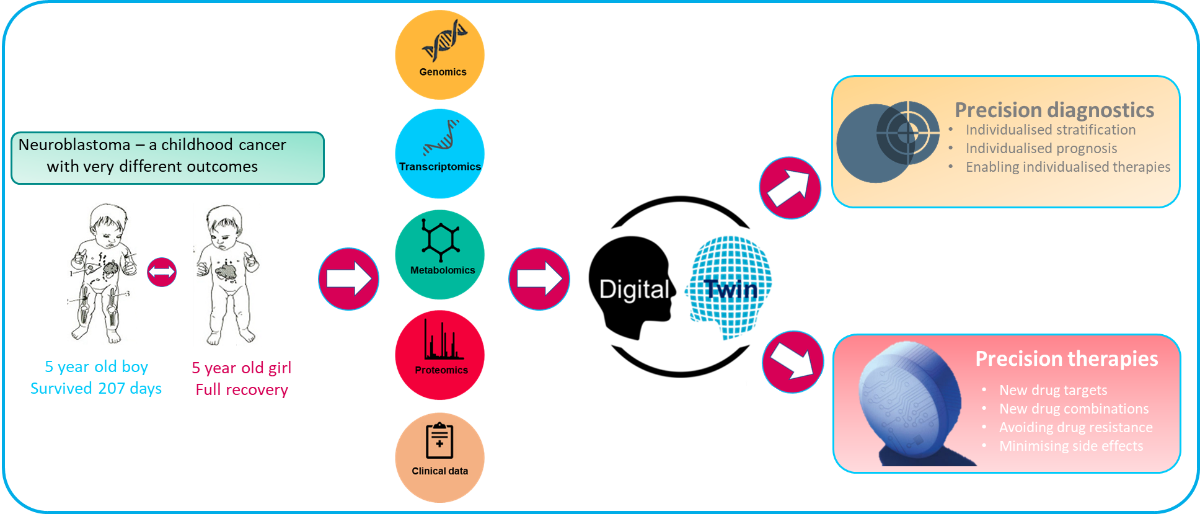
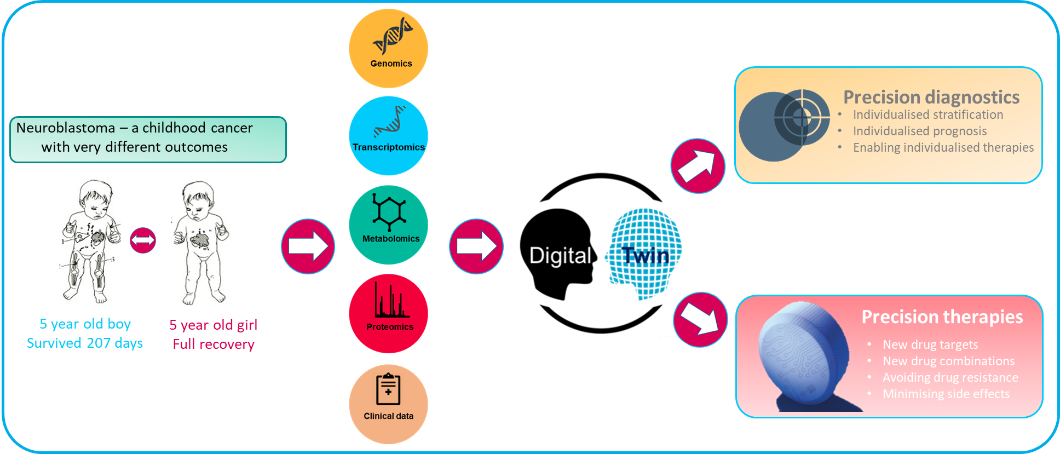
i) In order to enable a systematic search for potential targeted drugs that can replace cytotoxic chemotherapy, we have generated dynamic mathematical models that can predict both biochemical and biological responses. These models allow us to analyse the progression of the disease and potential therapy responses by in silico simulations which can be tailored to individual patients and enable us to find the best treatment for each patient. These predicted treatments can be compared with the clinically used treatments. It will take several rounds of stringent validation before the computational models can be used clinically, but we are aiming to achieve a proof of concept within the project lifetime.
ii) Our multi-omics pathway mapping in the EU funded ASSET project ((opens in a new window)https://cordis.europa.eu/project/id/259348) had indicated completely new targets for the therapy of high-risk NB, especially for those with amplification of the MYCN gene, which occurs in approximately 20% of NB patients. This gene amplification results in abnormally high concentrations of MYCN proteins. We found that this overexpresssed MYCN protein can regulate mitochondria. This regulation occurs on multiple levels and creates functional changes in the mitochondria that are unique to high risk NB cells with amplified MYCN genes. By characterizing these changes in molecular detail we aim to identify new drug targets that specifically target vulnerabilities of these aggressive cancer cells.
For further information, visit:
(opens in a new window)https://www.childrenshealthireland.ie/research-innovation-index/
Project team


Dr. Cormac Owens
Official Collaborator

Dr. Aleksandar Krstic
SBI Investigator

Melinda Halasz
Associate POI Member

Dr. Rashmi Sharma
Postdoctoral Researcher


