Promises of Tissue based imaging in Cancer research and Diagnostics
Transition to the Age of Digital Pathology
When a cancer patient first comes in contact with the clinician, a plethora of tasks sparks in their mind. These include proper diagnosis, selecting the best treatment option, monitoring treatment response, and lastly following up for recurrence of disease. To establish a diagnosis, biopsy tissue materials are often examined under the microscope by a histopathologist. In traditional practice, histopathologists examine glass slides of biopsy tissue under the microscope and characterise specific features relevant to the disease. Over recent years, cancer diagnosis has gone through a significant change due to a substantial improvement in computational power. Radical development of novel high-throughput imaging tools and image analysis algorithms has facilitated the gradual transition of manual histopathological analysis of traditional microscopy to digital pathology. Digital pathology involves digitising histopathological tissue slides into high-resolution images by scanning the slides with whole-slide scanners, and afterwards analysing the digital images with computer-aided image analysis software. After the slides are digitised to computer images, they can be viewed by a pathologist on a computer monitor, where the image can be magnified and navigated spatially in much the same way as standard microscopy. These scanned images represent the accurate depiction of the tissue slide, and in most cases, offer much more ease and flexibility in detecting complex morphological features of diagnostic or prognostic value. Image analysis software is then used to analyse those features of interest, and extract quantitative data from them that are generally much more accurate and reproducible. Figure 1 shows different steps in manual and digital pathology workflow.
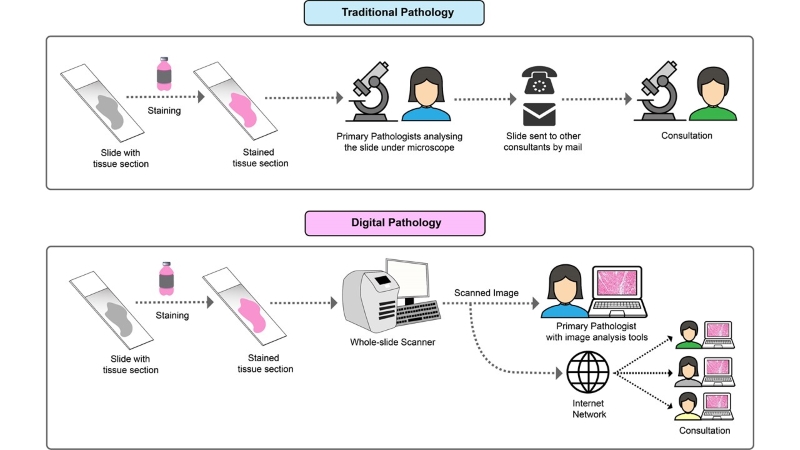
Figure 1: Comparison between the workflow of traditional and digital pathology
For visual assessment of the otherwise transparent tissue samples, the tissue sections go through staining procedures. There are various staining methods used for staining histopathological slides. For example hematoxylin and eosin (H&E) staining, which is used as the routine staining for histological slides, and provides a general overview of tissue morphology. But when pathologists or researchers need to look into more specified features such as presence or absence of specific molecular biomarker (protein/RNA) in cells, other specialised staining techniques like Immunohistochemistry (IHC) and In Situ Hybridization (ISH) are used. IHC uses targeted antibodies to detect a specific protein antigen and ISH uses complementary DNA or RNA strand to localise a particular nucleic acid sequence in the tissue. These antibodies and nucleic acids are normally linked with enzymes or fluorescent dyes, which makes the target visible under the microscope upon binding. Examples of scanned images of some tissue sections with IHC staining can be seen in the figure.
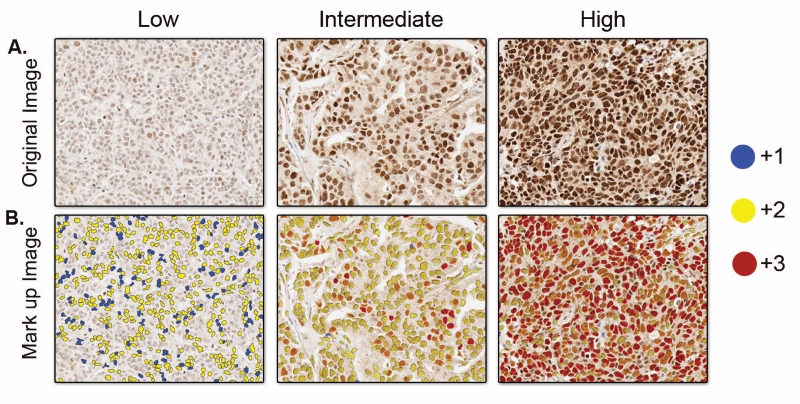
Figure 2: A. Scanned images of IHC staining on different tissue sections showing different levels of expression of a specific biomarker. More intensity of the staining indicates higher levels of the biomarker expression. B. Mark-up image with stained cells detected and annotated by an automated image analysis tool. Different colors of annotation indicates different threshold of staining intensity, blue (+1) indicates weak staining, yellow (+2) indicates moderate staining, and red (+3) indicates strong staining.
Based on these molecules linked with antibodies, there are two different types of IHC techniques: Chromogenic and Fluorescent. Chromogenic IHC depends on the chemical reaction triggered by the linked enzyme molecule and for fluorescent IHC, it’s the linked fluorescent dye. Traditionally, chromogenic IHC detects a single antigen in a slide. This can be a major limitation as cancer is a very complex molecular disease, and to diagnose or predict a proper treatment outcome, multiple molecular events or biomarkers need to be assessed. To solve this issue, Multiplexed Immunofluorescence (mIF) platforms have emerged as a potent tool as they allows simultaneous detection of multiple protein biomarkers on a single tissue section while preserving tumour material. It allows the opportunity to study different components of the tumour microenvironment, providing a better understanding of the cross-talk between the host and the tumour. While not yet used routinely in the clinic, this technique has the potential to dramatically improve diagnosis. Figure 3 graphically represents how multiple targets can be visualised simultaneously in the same tissue with multiplexing.
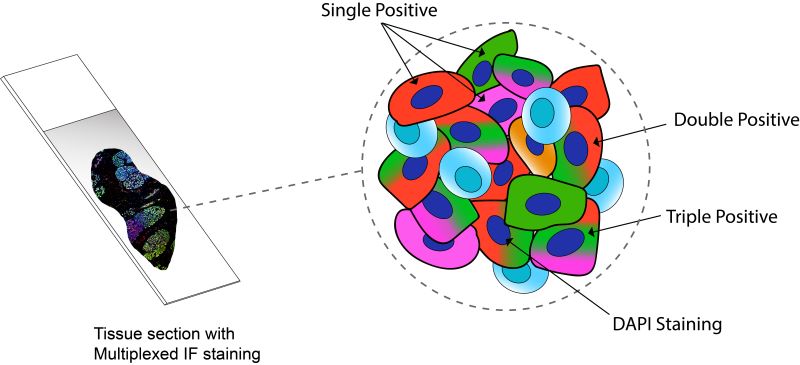
Figure 3: Tissue section with simultaneous staining of four different markers by Multiplexed Immunofluorescence. The schematic is a simplified representation of a magnified portion of the tissue showing cells expressing varied combinations of the three markers and a special fluorescence dye to light up all nuclei in the tissue (DAPI).
Deciphering Tumour Immune Contexture with Multiplex techniques
In recent years, there has been huge interest in the scientific community on mapping the immune architecture of tumours, and tissue-based multiplexed techniques have become one of the most desired tools for executing that. A tumour microenvironment hosts a highly diverse network of immune cell populations which collectively plays a vital role in tumour progression. A lot of different Immune therapies have been developed over the years which attempt to stimulate these immune cells to trigger a desired anti-tumour response, contrary to conventional cancer therapies that target tumour cells directly. Lately a good number of those therapies have also found major clinical success, although in a lot of cases it is still not properly understood why many patients do not respond to these treatments and/or experience immune-related complications. Recent research data suggest that tumour’s response to these immunotherapeutic approaches depends on the presence of specific immune cells in the tumour. These Immune cells surrounding tumour cells commonly referred to as tumour infiltrating lymphocytes (TIL) reflect the host’s immune response against the tumour cells. Numerous research studies have established that the composition of these TILs has significant deterministic value in disease prognosis for various cancer types like Breast cancer, Lung cancer, and colorectal cancer. Along with the presence of different immune cells in the tumour, there are other distinct features of TILs that have potential clinical significance. That is why characterisation or profiling of immune contexture in tumours requires detailed information about the tumour microenvironment, density of different immune cell populations, and their spatial arrangement. Here, spatial arrangement refers to the co-localisation of different immune cells in relation to the tumour cells and among themselves (Figure 4). In a recent study with non–small cell lung cancer, all these spatial features of TILs are found to have predictive value in the likelihood of the recurrence of cancer after chemotherapy. As proper characterisation of TILs demands multiple spatial features to be assessed together, multiplexed staining platforms have emerged as an ideal tool for immune profiling in cancer research. In contrast to other tissue-based imaging tools, multiplexed platforms are the only platforms that allow studying the spatial distribution of multiple immune and non-immune biomarkers and the interactions among them.
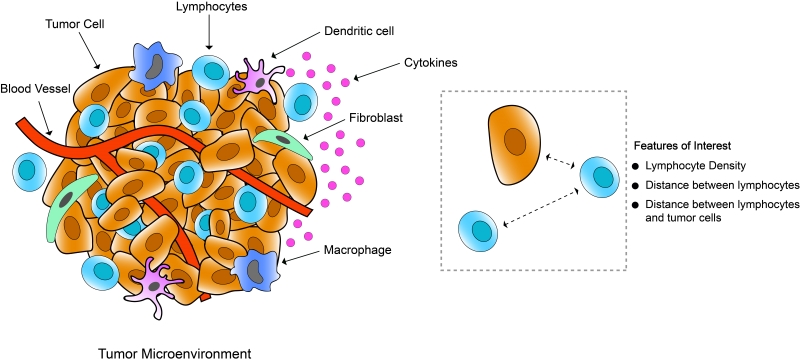
Figure 4: Schematic representation of tumour microenvironment showing tumour infiltrating lymphocytes, epithelial cells, fibroblasts, blood vessels, other immune cells (macrophages, dendritic cells).
Incorporation of Artificial Intelligence in Digital Pathology
Along with the use of high-end imaging tools in clinical and research practice, the need for increasing translational outcome of these tissue-based biomarkers have continued the innovation of new and superior image analysis approaches. Over time in tissue-based biomarker analysis, more and more complex and sub visual features are being explored in clinical practice and academic research. Moreover, the enormous amount of data being generated from these advanced imaging tools has made management of diagnostic workflows increasingly tough. That is why there’s an increasing need to develop tools for pathologists to help them in achieving significant growth in clinical outcome. Artificial Intelligence (AI) comes right into play here. In simple terms, AI is machine-based approaches that emulate human intelligence; its purpose is to try making predictions like an intelligent human does. Integration of AI in image analysis of tissue-based biomarkers offers far more efficient and robust extraction of clinically relevant data from a large patient population and reduces human error to a large extent. It makes a subjective assessment of images quantifiable and extremely reproducible. In recent times, AI approaches for image analysis are shifting towards Deep Learning, which is based on artificial neural networks that imitate the learning process of the human brain. The way deep learning works in pathology is that pathologists feed the network with annotations of a feature in an image (e.g. – blood vessel), and then the neural network learns from those annotations like a human does and later tries to detect those features by itself in the tissue specimen. The more input data it gets, the more precise it gets at identifying the feature of interest and found to match or in many cases perform even better than humans.
There are still quite some challenges to integrating AI in regular clinical diagnostics exclusively. Nevertheless, the potential is boundless. We are in an age where treatment strategies are more and more being tailored to individual patients. Since advanced tissue-based imaging technologies offer ways of acquiring and assessing patient-specific clinical data with far more accuracy and precision than usual, these technologies are now growing immensely in popularity and demand in histology labs, hospitals, universities, and pharmaceutical companies worldwide. Many of them are leaning towards digitising their entire workflow. Over the next ten years, progress in this field will undoubtedly transform the field of oncology and related medical diagnostics. Ultimately, given the phenomenal rise of machine learning and AI, one can only imagine the sensational advancements that will be uncovered in the field of medicine in the next decade.
Author: Chowdhury Arif Jahangir, POI PhD student
 After completing a Master's degree in Molecular Biotechnology under a joint program by Karolinska Insititute and University of Skövde (Sweden), Arif received a research assistantship at Karolinska institute. There Arif worked on investigating the molecular mechanism behind antioxidant induced lung cancer metastasis. After completing the project, Arif joined the Cancer Biology and Therapeutics Lab at UCD as a research assistant and worked under the OPTi-PREDICT project funded by SFI. Arif received his 1st class undergraduate degree (BSc) in Biotechnology from the University of Rajshahi (Bangladesh).
After completing a Master's degree in Molecular Biotechnology under a joint program by Karolinska Insititute and University of Skövde (Sweden), Arif received a research assistantship at Karolinska institute. There Arif worked on investigating the molecular mechanism behind antioxidant induced lung cancer metastasis. After completing the project, Arif joined the Cancer Biology and Therapeutics Lab at UCD as a research assistant and worked under the OPTi-PREDICT project funded by SFI. Arif received his 1st class undergraduate degree (BSc) in Biotechnology from the University of Rajshahi (Bangladesh).
In May 2020, Arif started his Ph.D. with POI, under the supervision of Professor William Gallagher. His project aims to explore the interplay between master transcriptional regulators and immune cells within the tumor microenvironment of breast cancer. His project is co-funded by the Irish Cancer Society.

