

P2.5: Targeting the XBP1s biogenesis pathway in cancer
Project Overview
The endoplasmic reticulum (ER) stress response signalling pathway is important in cancer, and increasingly big pharma are looking for drug targets in this pathway. One arm of this is the IRE1/XBP1s signalling pathway which contributes to cell survival during ER stress. Its constitutive activity indicates poor prognosis in several cancer types. To date, targeting of this pathway has focused on inhibiting IRE1. We have shown that inhibiting IRE1 enzyme activity enhances response to chemotherapy in triple-negative breast cancer (TNBC) [1]. However, IRE1 activates two downstream pathways, one leading to production of the XBP1s transcription factor and the other leading to the regulated IRE1-dependent decay (RIDD) of several RNAs. XBP1s high/RIDD low tumours are most aggressive, highlighting the potential benefits of targeting the XBP1s branch of IRE1 activity rather than IRE1 itself. In this regard, the RNA ligase HSPC117 is an attractive target, since its inhibition would selectively block the production of XBP1s without affecting RIDD. No such drug molecules are currently available.
This project aims to design and develop small molecules targeting HSPC117 to selectively inhibit the XBP1s arm of IRE1 downstream signalling in cancer. We have shown that knockdown of XBP1s affects the growth of TNBC tumours, underscoring the validity of our hypothesis. To design the inhibitors we have performed structure-based drug design using the homology-based model of HSPC117 [2]. The most promising drug candidates were initially tested biochemically and in cancer cell lines to determine their toxicity, their ability to sensitise cancer cells to cell death by chemotherapeutic drugs, and their effect on cancer cell migratory capacity and invasiveness. The lead compound with the best profile will be tested in preclinical models.
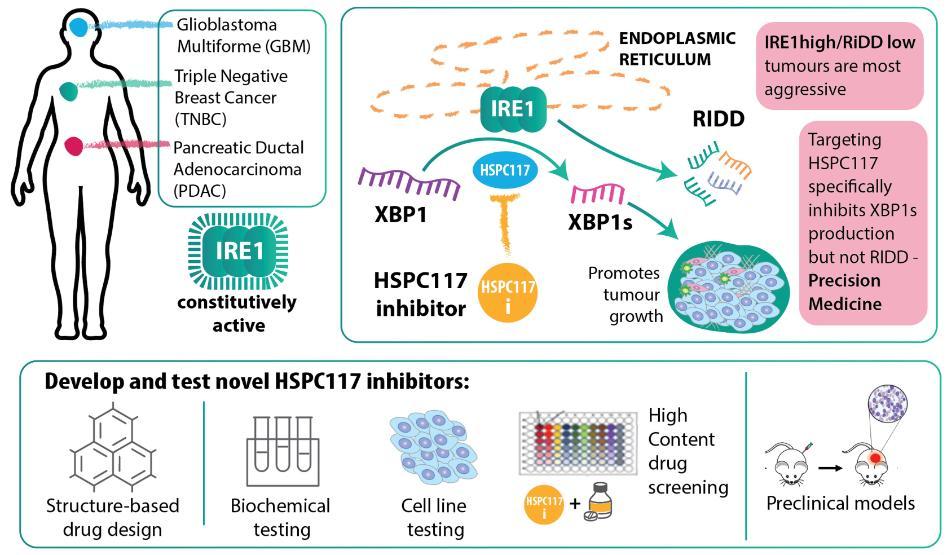

Targeting HSPC117 offers a potentially more precise way to exploit ER stress signalling for cancer therapy. The industry partner Cell Stress Discoveries has excellent opportunities to grow this area due to its unique expertise and collaborations with leading ER stress experts within POI. The development of HSPC117 inhibitors will enable precise targeting of cancer and lead to novel combination therapies.
For further information, visit
(opens in a new window)https://cellstressdiscoveries.com/
Key References:
1. Logue S et al. "Inhibition of IRE1 RNase activity modulates the tumor cell secretome and enhances response to chemotherapy." Nature Communications 9.1 (2018): 1-14.
2. Nandy A et al. "Homology model of the human tRNA splicing ligase RtcB." Proteins: Structure, Function, and Bioinformatics 85.11 (2017): 1983-1993.
Project team



