Plain Language Statement
Cancer cachexia (kuh-KEK-see-uh) is a muscle-wasting disorder characterised by weight loss, inflammation, loss of appetite, and reduced treatment tolerance. It affects up to 80% of cancer patients and is responsible for the death of around 20% of these patients. Current treatment options are limited and mainly target symptoms, for example, anti-nausea medication and appetite stimulants. However, there is hope. The drug Anamorelin, developed by the Swiss pharma company Helsinn, will address poor appetite and the resulting malnutrition in cachexia patients. Anamorelin works by mimicking ghrelin, the hunger hormone in your body, to increase appetite, by enhancing food intake, then improve nutrition status and muscle mass. It has recently been approved for use in Japan for the treatment of cachexia. However, results in the clinical trials were not as clear-cut as they hoped. And so, it is our goal to develop a biomarker, or indicator, of health and cachexia status to tell us who will benefit from the drug Anamorelin.
To develop this biomarker, we are using High-Density Lipoprotein (HDL), sometimes termed the “good” cholesterol within the blood, for proteomic analysis. Proteomics tells us what proteins are attached to the HDL, called the proteome. This provides us with a large amount of information in relation to the body’s processes. The HDL proteome is highly reflective of inflammation and body composition, both important in relation to cachexia. We hope to find a pattern of proteins only seen in cachexia patients, which will allow us to identify others who may have the disease. To begin this project, we will first look for a biomarker of sarcopenia, the muscle loss associated with old age, in a group of participants over the age of 65. This is to determine how well we can develop a biomarker from HDL in relation to muscle loss and also allow us to better distinguish between those with sarcopenia and cachexia in our cancer group.
Project Overview
Diet is an established environmental factor that may play a role in cancer risk and progression. Nutritional components can drive obesity-related metabolic and inflammatory pathways that can impact cancer development. Nutrition is an essential adjunct to effective cancer therapy, and can greatly impact cancer risk. Firstly, it can impact potential triggers or drivers of obesity-related cancers. Secondly, malnutrition is a key characteristic of ongoing disease, wherein cancer-associated cachexia (CAC) is very common.
CAC is the ongoing loss of appetite resulting in a reduction in both skeletal muscle mass, and adipose tissue which affects up to 80% of cancer patients. CAC severely compromises patient’s quality of life, treatment tolerance, nutritional status and is responsible for approximately 20% of cancer deaths. As the growing tumour energy demands increase, there is a disturbance in protein metabolism, along with inflammation that leads to muscle atrophy. Cancer treatment also negatively effects energy intake due to nausea, diarrhoea, mucositis and changes in taste perception. Cachexia worsens malnutrition by increasing metabolism and protein synthesis and restricting energy and nutrient intake, leading to deficiencies such as anaemia and contributing to muscle loss. Inflammation further worsens malnutrition and cachexia by interfering with the hunger regulating hormones leptin and ghrelin, disrupting metabolic homeostasis.
Current therapies for this condition are very poor, but a promising approach is to combine a nutritional intervention strategy with a drug called a ghrelin agonist. Ghrelin agonist drugs can have multiple effects on the body, including increasing appetite and lean body mass and decreasing energy expenditure.
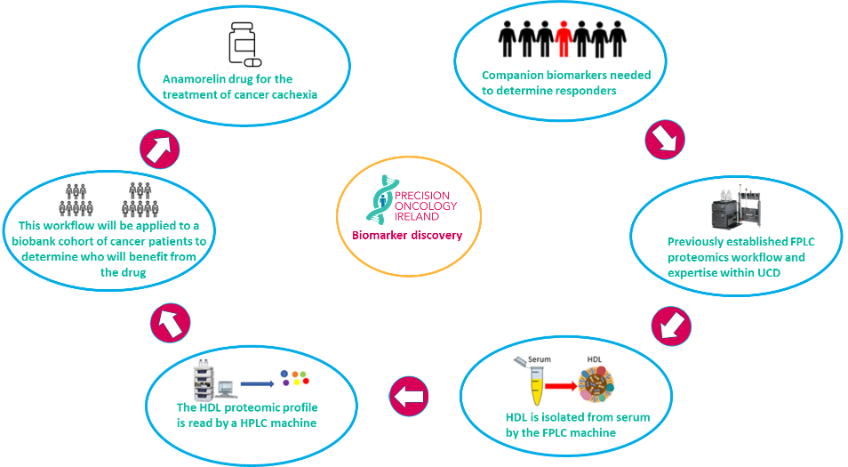
Part of this POI Nutrigenomics project is focused on interactions with a therapy called Anamorelin which is produced by the partner company Helsinn and is not currently approved for cachexia treatment. This drug has undergone clinical trials in non-small cell lung cancer, which has a high incidence of cachexia and associated shortened survival. While some patients showed increased body weight and improved quality of life, responses varied considerably between patients. Therefore, this project aims to develop reliable companion biomarkers to identify which patients are most likely to benefit from Anamorelin therapy. The research will focus on the use of exosome and plasma high-density lipoprotein (HDL) proteomic signatures and will integrate this information with clinical data and the nutritional environment using Nuclear Magnetic Resonance metabolomics. The HDL proteome has been well characterised in other inflammatory diseases such as cardiovascular disease. We suspect that the HDL proteome will be a good area for biomarker discovery as it is greatly affected by nutritional and inflammatory status. The outcome of this project will provide a more complete understanding of both dietary impacts and cachexia during cancer development and progression, with the proposed biomarker-based diagnostic test allowing for individual patient stratification for treatment and monitoring of treatment response.
Secondly, we will determine the impact of different dietary elements on potential cancer risk e.g. saturated versus unsaturated fats on inflammation. This paradigm is probably more relevant within the context of obesity-related cancers. A more complete understanding of if and how nutrition impacts on metabolic and inflammatory profiles will advance knowledge so that we can maximise the efficacy of potential diet-drug interventions.
Ultimately POI will enable us to understand if and how nutritional intake can alter metabolism and inflammation, two key elements in cancer biology, which undoubtedly interact with drug therapy efficacy.
Project Team

Helen Roche
Project Lead
Kathleen Mitchelson
PhD student

